PVP K60
5 USD ($)
Product Details:
- Taste Tasteless
- Heavy Metal (%) 0.002%
- Loss on Drying 5.0%
- Smell Odorless
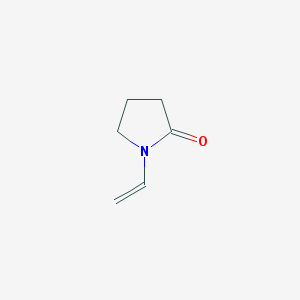
- Structural Formula (C6H9NO)n
- Molecular Weight Approx. 360,000
- Storage Keep container tightly closed in a dry and well-ventilated place.
- Click to View more
X
PVP K60 Price And Quantity
- 100 Kilograms
- 5 USD ($)
PVP K60 Product Specifications
- 3.0-7.0 (5% aqueous solution)
- >99%
- (C6H9NO)n
- 9003-39-8
- White to off-white powder
- Polyvinylpyrrolidone K60
- Below 80 mesh
- 39059990
- Powder
- PVP K60
- 618-340-6
- White to off-white
- Powder
- 0.002%
- 5.0%
- Odorless
- Tasteless
- (C6H9NO)n
- Approx. 360,000
- Keep container tightly closed in a dry and well-ventilated place.
- 2 Years
- Pharmaceutical Grade
- Pharmaceutical and Cosmetic Application
- Soluble in water and many organic solvents
- Non-poisonous
- 160C (decomposes)
- 25 kg drum or as required
- Positive reaction for polyvinylpyrrolidone
- 400 ppm
- < 0.5 EU/mg
- 0.04%
- 0.1%
- Total bacterial count < 1000 cfu/g
- 0.25-0.35 g/ml
- 55.0-65.0
PVP K60 Trade Information
- WHOGM ,COA ,DMF
Product Description
Providone is also known as polyvinylpyrrolidone (PVP) or polyvidone which is a synthetic water-soluble polymer made from the monomer N-vinylpyrrolidone. It is used as a binder in many pharmaceutical tablets and used in many technical applications with various roles such as an adhesive, additive, and emulsifier. It is also used in some eye drops as a lubricant. PVP added to iodine forms a complex called povidone-iodine that possesses disinfectant properties due to the presence of iodine, a bactericidal component. Providone-iodine can be found in solutions, ointment, pessaries, liquid soaps and surgical scrubs.
Versatile Applications in Pharmaceuticals and Cosmetics
PVP K60 serves as a key ingredient in numerous pharmaceutical and cosmetic products, valued for its film-forming, binding, and stabilizing characteristics. Its purity, solubility, and safety profile make it suitable for tablets, ointments, creams, and other formulations, helping manufacturers achieve consistent product quality and efficacy.
Superior Quality and Safety Standards
This grade of polyvinylpyrrolidone adheres to strict safety limits, with low residual monomers and heavy metals, and a microbial count less than 1000 cfu/g, ensuring compatibility with sensitive pharmaceutical and cosmetic applications. Its identity, absence of odor, and high purity further emphasize its reliability for industrial use.
FAQs of PVP K60:
Q: How can PVP K60 be used in pharmaceutical and cosmetic formulations?
A: PVP K60 acts as a binder, film-former, and stabilizer in tablet production, creams, and gels. Its excellent solubility and safety features make it suitable for both oral and topical applications.Q: What benefits does PVP K60 offer over other grades of polyvinylpyrrolidone?
A: With a K-value of 55.065.0, PVP K60 provides balanced viscosity and superior binding properties, alongside high purity and compliance with stringent microbial and heavy metal limits, making it ideal for pharma-grade applications.Q: When should manufacturers consider using PVP K60 in their formulations?
A: Manufacturers should opt for PVP K60 when high purity, controlled viscosity, strong binding, and reliable safety compliance are required, such as in pharmaceutical tablets, topical medicines, and premium cosmetics.Q: Where can PVP K60 be stored to maintain its stability?
A: PVP K60 should be kept in tightly closed containers in dry, well-ventilated areas to preserve its physical integrity and extend its shelf life for up to two years.Q: What process is recommended for handling and dissolving PVP K60?
A: PVP K60 can be easily dissolved in water or many organic solvents. It is recommended to disperse the powder gradually under agitation to achieve a homogenous solution for optimal use in formulations.Q: Is PVP K60 safe for use in sensitive formulations?
A: Yes, PVP K60 is non-poisonous, tasteless, and odorless, with low residual monomers and heavy metals, plus minimal endotoxin levels. These characteristics render it safe for sensitive pharmaceutical and cosmetic preparations.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email